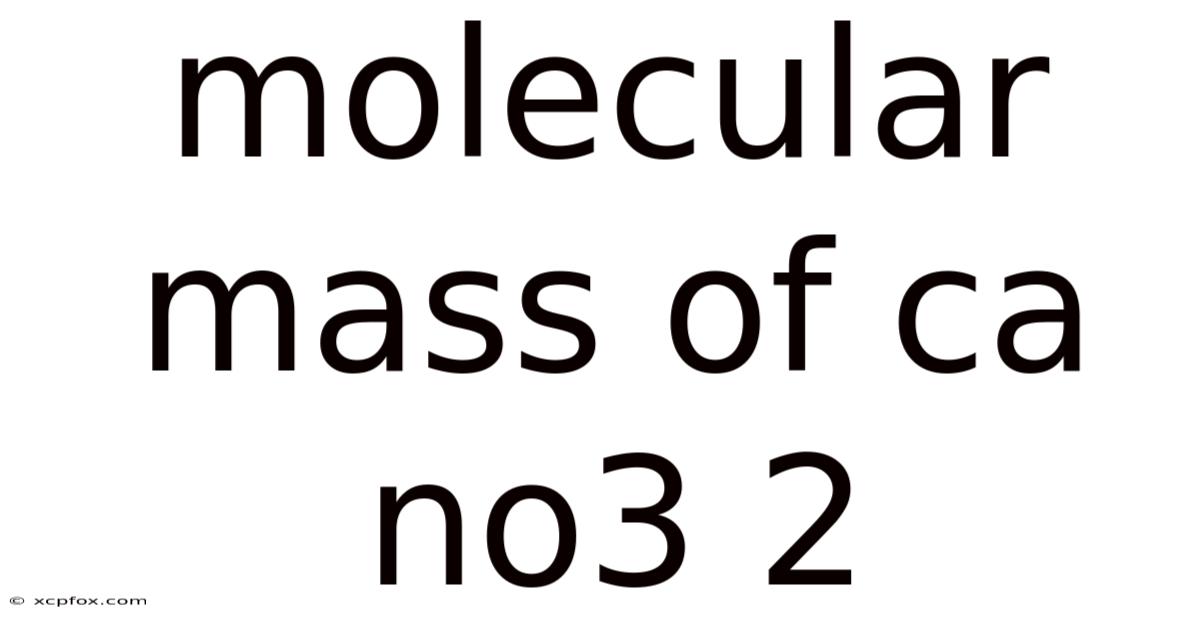Imagine you're in a chemistry lab, meticulously weighing out reactants for an experiment. Every calculation, every measurement hinges on understanding the fundamental properties of the substances you're working with. One such crucial property is molecular mass—a concept that ties together the microscopic world of atoms and molecules with the macroscopic world of grams and kilograms we experience daily.
Think of molecular mass as the weight of a single molecule. It's not something you can directly measure on a scale, but rather a calculated value derived from the atomic masses of all the atoms within that molecule. In real terms, it's the key to converting between mass and moles, enabling precise control over chemical reactions and processes. Mastering the concept of molecular mass unlocks a deeper understanding of chemical stoichiometry and quantitative analysis. In this comprehensive exploration, we'll dive into the intricacies of calculating the molecular mass of Calcium Nitrate, Ca(NO3)2, and explore its significance in various fields.
Molecular Mass of Ca(NO3)2: A thorough look
The molecular mass of a compound is the sum of the atomic masses of all the atoms in the molecule. And to accurately determine the molecular mass of Calcium Nitrate, Ca(NO3)2, we must first understand its chemical formula and the atomic masses of its constituent elements: calcium (Ca), nitrogen (N), and oxygen (O). Which means understanding these elements and their roles is foundational to grasping the broader implications of Calcium Nitrate in various chemical and industrial processes. This detailed guide will walk you through the steps to calculate the molecular mass of Ca(NO3)2, explore its importance, and discuss its practical applications Worth keeping that in mind..
Comprehensive Overview
Definitions and Foundational Concepts
Molecular mass, often expressed in atomic mass units (amu) or grams per mole (g/mol), is the mass of a single molecule. Which means it is calculated by summing the atomic masses of each atom present in the molecular formula. Atomic mass, on the other hand, is the mass of a single atom, typically found on the periodic table. These atomic masses are determined experimentally and are based on the mass of carbon-12 isotope, which is defined as exactly 12 amu.
The concept of molecular mass is closely tied to the mole concept, which is a fundamental unit in chemistry used to express the amount of a substance. One mole is defined as the amount of substance that contains as many elementary entities (atoms, molecules, ions, etc.) as there are atoms in 12 grams of carbon-12. This number, known as Avogadro's number (approximately 6.022 x 10^23), provides a bridge between the microscopic world of atoms and molecules and the macroscopic world that we can measure and observe Worth keeping that in mind. Practical, not theoretical..
Scientific and Historical Context
The development of the concept of molecular mass is rooted in the early 19th-century work of scientists like John Dalton, who proposed the atomic theory of matter. Dalton's theory suggested that all matter is composed of indivisible particles called atoms, and that chemical compounds are formed by the combination of atoms in simple whole-number ratios. This significant idea laid the foundation for understanding the quantitative relationships in chemical reactions.
Later, scientists like Amedeo Avogadro and Stanislao Cannizzaro made significant contributions to determining the relative masses of atoms and molecules. Avogadro's hypothesis, which stated that equal volumes of gases at the same temperature and pressure contain equal numbers of molecules, was crucial for establishing a consistent scale of atomic masses. Cannizzaro's work helped resolve many of the inconsistencies and ambiguities in the existing atomic mass data, paving the way for the modern periodic table and a more accurate understanding of molecular masses.
Calculation of Molecular Mass of Ca(NO3)2
To calculate the molecular mass of Calcium Nitrate, Ca(NO3)2, we need to know the atomic masses of each element involved:
- Calcium (Ca): Approximately 40.Here's the thing — 08 amu
- Nitrogen (N): Approximately 14. 01 amu
- Oxygen (O): Approximately 16.
The chemical formula Ca(NO3)2 indicates that each molecule of Calcium Nitrate contains one calcium atom, two nitrogen atoms (inside the parenthesis), and six oxygen atoms (3 oxygen atoms multiplied by 2).
Because of this, the molecular mass of Ca(NO3)2 is calculated as follows:
Molecular Mass of Ca(NO3)2 = (1 x Atomic Mass of Ca) + (2 x Atomic Mass of N) + (6 x Atomic Mass of O) Molecular Mass of Ca(NO3)2 = (1 x 40.08 amu) + (2 x 14.Because of that, 00 amu) Molecular Mass of Ca(NO3)2 = 40. 01 amu) + (6 x 16.08 amu + 28.02 amu + 96.00 amu Molecular Mass of Ca(NO3)2 = 164 And it works..
This is where a lot of people lose the thread.
That's why, the molecular mass of Calcium Nitrate, Ca(NO3)2, is approximately 164.10 amu. This value can also be expressed as 164.10 g/mol when dealing with molar quantities And it works..
Importance of Molecular Mass
The molecular mass is a critical parameter in various chemical calculations and applications. It serves as a bridge between mass and moles, enabling chemists and scientists to convert between these two fundamental units. Knowing the molecular mass of a compound allows for precise control over chemical reactions, ensuring that reactants are combined in the correct stoichiometric ratios.
In analytical chemistry, molecular mass is essential for determining the concentration of solutions and quantifying the amount of a substance in a sample. It is also crucial in the synthesis of new compounds, where precise control over the amounts of reactants is necessary to achieve the desired product yield and purity.
Not the most exciting part, but easily the most useful Most people skip this — try not to..
Applications of Calcium Nitrate
Calcium Nitrate, Ca(NO3)2, is a versatile chemical compound with numerous applications across various industries. One of its primary uses is in agriculture, where it serves as a valuable fertilizer. It provides plants with essential nutrients, such as nitrogen and calcium, which are crucial for healthy growth and development. Calcium Nitrate is particularly effective for crops grown in acidic soils, as it helps to neutralize the soil pH and improve nutrient availability Surprisingly effective..
Not the most exciting part, but easily the most useful.
In wastewater treatment, Calcium Nitrate is used to control odors and prevent the formation of hydrogen sulfide (H2S), a toxic and corrosive gas. It promotes the growth of beneficial bacteria that convert H2S into less harmful compounds, thereby improving the quality of the treated water.
Calcium Nitrate also finds applications in the construction industry, where it is used as an additive in concrete to accelerate the setting time and improve its durability. It helps to reduce the risk of corrosion of steel reinforcement bars embedded in concrete, extending the lifespan of structures.
Trends and Latest Developments
Current Trends in Fertilizer Use
The agricultural industry is increasingly focused on sustainable and efficient fertilizer practices. Consider this: traditional nitrogen fertilizers, such as urea and ammonium nitrate, can contribute to environmental problems like greenhouse gas emissions and water pollution. So naturally, there is growing interest in alternative nitrogen sources, including Calcium Nitrate, which is considered to be more environmentally friendly due to its lower volatility and reduced risk of nitrogen losses Simple, but easy to overlook. Still holds up..
Precision agriculture, which involves using data and technology to optimize crop management practices, is also gaining traction. Day to day, this approach allows farmers to tailor fertilizer applications to the specific needs of their crops, reducing waste and minimizing environmental impacts. Calcium Nitrate, with its readily available nitrogen and calcium, is well-suited for precision agriculture applications Practical, not theoretical..
It sounds simple, but the gap is usually here.
Innovations in Wastewater Treatment
The use of Calcium Nitrate in wastewater treatment is also evolving. Researchers are exploring new ways to enhance its effectiveness in controlling odors and preventing H2S formation. One promising approach involves combining Calcium Nitrate with other additives, such as iron salts or bioaugmentation cultures, to further improve the treatment process.
Additionally, there is growing interest in recovering valuable resources from wastewater, such as nitrogen and phosphorus. Calcium Nitrate can play a role in these recovery processes by facilitating the precipitation of nutrients as recoverable solids.
Advances in Concrete Technology
In the construction industry, Calcium Nitrate is being used in conjunction with other additives and technologies to produce high-performance concrete. Self-healing concrete, which can automatically repair cracks and damage, is one such innovation. Calcium Nitrate can be incorporated into self-healing concrete formulations to promote the formation of calcium carbonate, which helps to seal cracks and prevent further deterioration.
On top of that, there is ongoing research into the use of Calcium Nitrate as a corrosion inhibitor in concrete exposed to harsh environments, such as marine structures and bridges. Its ability to reduce the risk of steel corrosion makes it a valuable tool for extending the lifespan of these critical infrastructure assets.
Tips and Expert Advice
Maximizing the Benefits of Calcium Nitrate in Agriculture
To maximize the benefits of Calcium Nitrate as a fertilizer, it is the kind of thing that makes a real difference. Soil testing can help determine the specific nutrient needs of the crop, allowing for a tailored fertilizer application. It is also important to consider the soil pH, as Calcium Nitrate is most effective in slightly acidic to neutral soils.
Not the most exciting part, but easily the most useful.
Applying Calcium Nitrate in split applications, rather than a single large dose, can help to improve nutrient uptake and reduce the risk of leaching. Foliar application, where the fertilizer is sprayed directly onto the leaves of the plant, can also be an effective way to deliver nutrients quickly and efficiently.
Optimizing Calcium Nitrate Use in Wastewater Treatment
When using Calcium Nitrate in wastewater treatment, it is the kind of thing that makes a real difference. Calcium Nitrate works best in slightly alkaline conditions and in the presence of sufficient dissolved oxygen. Regular monitoring of these parameters can help confirm that the treatment process is operating optimally.
It is also important to consider the dosage of Calcium Nitrate. Overdosing can lead to the accumulation of nitrate in the treated water, which can be harmful to aquatic life. Underdosing, on the other hand, may not be effective in controlling odors and preventing H2S formation That's the part that actually makes a difference..
Ensuring Proper Handling and Storage of Calcium Nitrate
Calcium Nitrate is a relatively stable compound, but it should be handled and stored properly to prevent accidents and ensure its quality. Practically speaking, it should be stored in a cool, dry place, away from direct sunlight and heat sources. It should also be kept away from combustible materials, as it can act as an oxidizer.
When handling Calcium Nitrate, it is important to wear appropriate personal protective equipment, such as gloves and eye protection. Avoid breathing dust or fumes, and wash thoroughly after handling. In case of accidental ingestion or contact with skin or eyes, seek medical attention immediately Simple, but easy to overlook..
FAQ
Q: What is the difference between molecular mass and molar mass? A: Molecular mass refers to the mass of a single molecule and is expressed in atomic mass units (amu), while molar mass refers to the mass of one mole of a substance and is expressed in grams per mole (g/mol). Numerically, they are the same, but they differ in units and the quantity they represent.
Q: Is Calcium Nitrate an organic or inorganic compound? A: Calcium Nitrate is an inorganic compound. Organic compounds are generally defined as those containing carbon-hydrogen bonds, which are not present in Calcium Nitrate Still holds up..
Q: How does Calcium Nitrate affect soil pH? A: Calcium Nitrate can help to neutralize acidic soils by providing calcium ions, which react with acidic components in the soil That's the part that actually makes a difference..
Q: Can Calcium Nitrate be used in hydroponics? A: Yes, Calcium Nitrate is commonly used in hydroponics as a source of calcium and nitrogen for plants.
Q: Is Calcium Nitrate safe for human consumption? A: While Calcium Nitrate is used as a fertilizer and in some food applications, it is not safe for direct human consumption in large quantities. It should be handled with care and used according to recommended guidelines.
Conclusion
Understanding the molecular mass of Calcium Nitrate, Ca(NO3)2, is fundamental to grasping its role in various applications, from agriculture to wastewater treatment and construction. By calculating the molecular mass (approximately 164.10 amu), we can accurately determine the amounts of Calcium Nitrate needed for specific applications, ensuring optimal results and minimizing waste.
This practical guide has provided insights into the definitions, historical context, and practical uses of Calcium Nitrate. By following the tips and expert advice outlined, you can maximize its benefits while ensuring safe handling and storage.
Now, it's your turn to take action. But whether you're a student, researcher, or industry professional, deepen your understanding of Calcium Nitrate by exploring its applications in your field of interest. So share your findings and insights with others, and contribute to the ongoing efforts to optimize its use for a more sustainable future. Start a discussion in the comments below and let us know how you are applying the knowledge of the molecular mass of Ca(NO3)2 in your work or studies The details matter here. Which is the point..